 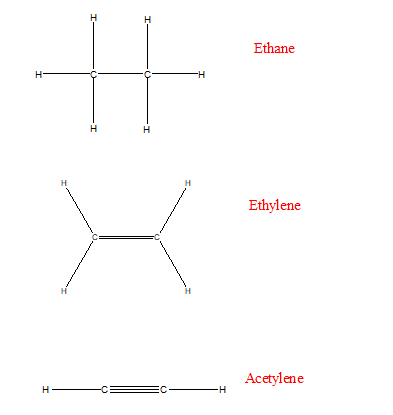 
The classic example of the Lewis structure breaking down is the dioxygen molecule, which is often written with a double bond as a Lewis structure but experimental evidence indicates that there are two unpaired electrons involved in bonding. If a Lewis structure shows an "expanded" octet (or for transition metal complexes), you are better off looking at a molecular orbital diagrams to determine what the bond order is and whether the bond in question is sigma or pi (whether there is a node along the bond axis). This assumes that the Lewis structure is a good representation of the molecule. triple bond: one sigma and two pi bonds.For multiple bonds, one is a sigma bond and the remainder are pi bonds. So, my doubt was focused on that aspect.Ī single bond in a Lewis structure is considered a sigma bond. But, if I am given a molecule (such as acetylene, ozone or ammonia) I don't know how to apply the Theory correctly, I get confused when it is a sigma link or pi link. Starting with its Lewis structure, the C2F2. I also understand the concept of hybridization and overlapping of atomic orbitals (simply the algebraic combination of wave functions). What is the molecular geometry of C2F2 The molecular shape around each carbon atom of the C2F2 molecule is linear. I know how to apply the VSEPR model: I determine the Lewis structure and observe how many electron pairs the central atom has. In an exam, you just have to hope that the examples given work well for the level of theory (or the rules of thumb) you are applying. Also, the predicted angles mostly will be approximations (except in the case of high symmetry, e.g. The predictions work pretty well for some compounds made of H, C, N, O atoms, less so for third row main group elements, and they mostly break down for transition metals. You would measure or deduce the bond lengths and angles with an appropriate experiment, and see whether your data fits your prediction. How do I know what's wrong with VSEPR theory a priori? For example, in an exam I am asked to determine this structure, how to do it so that it is always the right one. So a triple bond in the Lewis structure is one sigma and two pi bonds in the valence bond formalism. It helps with determining polarity, phase of matter, magnetism, reactivity, color, and biological activity of a molecule, in short, anything and everything about a molecule can be studied through molecular geometry. The Lewis structures are a way to illustrate the valence bond description as far as possible. Molecular Geometry of Acetylene (C2H2) Studying the molecule geometry of a molecule is a fundamental step in chemistry to analyze the behavioral properties of any molecule. This is where I don't know how to see if the link is σ- or π-type. In this case, there are two central atoms: $\ce$, which is expected to be linear.įor a complete study, I applied the valence bond model based on the hybridization of the atomic orbitals. To predict molecular geometry I used the VSEPR model, which is based on the number of electron pairs in the central atom to determine molecular geometry. For this I performed the following procedure.įirst, I drew the correct Lewis structure which is a graphical representation that shows the pairs of electrons linking the atoms of a molecule and the pairs of solitary electrons which may exist. I need to determine the molecular geometry of acetylene.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
